Soap 101 Making Lye
|
A Timeline of Soap Making (from Soap Making Resource) 2200 B.C. - Although it is believed by many that soap's history actually began around 2800 B.C. in Babylonia (where historians found a barrel containing a soap-like substance) unarguable proof of soaps existence was first provided by a 2200 B.C. Mesopotamian clay tablet with an actual soap recipe inscribed on it. This ancient soap making technique described mixing potash and oils to form a cleaning agent. 1500 B.C. - Egyptian manuscripts describe a matter created by combining animal fats and vegetable oils to create a soap-like substance. The ancient documents go on to explain another type of soap that is used in the production of wool. 200 A.D. - The very first allusion to soap making in literature occurred by a famous Greek physician named Galen. 600 A.D. - soap making guilds were formed and the modern formula for soap that we use today was created. 900 A.D. - soap started to be produced commercially and sold for .3 Dinars per bar. c1700-c1800 A.D. - During the industrial revolution, soap began its production in factories and stopped being produced in the home kitchen. Currently - Soap is being produced in the same way as it was centuries ago after the industrial revolution. As almost all people in this modern age purchase and use commercially manufactured soaps, the history of soap making is at somewhat of a standstill.
What is Soap? By chemical definition, soap is the salt of a fatty acid, a triglyceride, which are three molecules of fatty acids attached to a single molecule of glycerol (glycerin). The bars we use for cleaning are simply animal or vegetable oils treated with a strong alkaline solution called lye (sodium hydroxide). Lye catalyzes the saponification process, the hydrolysis of fats into free fatty acids, which then combine with alkali to form crude soap. The liberated glycerin by-product is either left in the soap or collected, depending on which soap-making process is used.
|
| What is Lye? | |
| There is a difference between
homemade, or original lye, and commercial lye. |
|
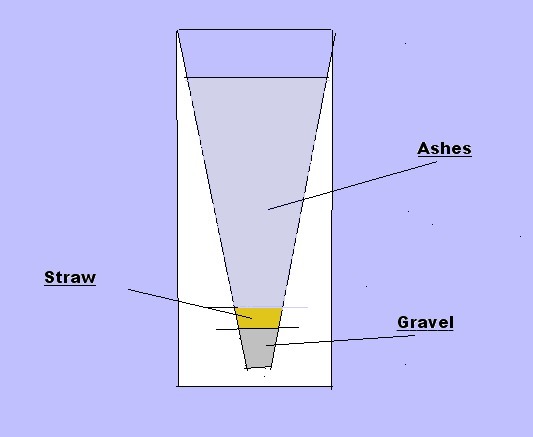
| Homemade
lye is Potassium Hydroxide and can be made
rather simply at home from wood ashes. Potassium Hydroxide makes a softer soap, so the fat or oils used will need to be beef tallow (beef fat) or even lard (pig fat). These are 'harder' fats. You can use other fats, but the finished product will be much softer. |
|
| Commercial lye is
Sodium Hydroxide,
which is much more complex to manufacture, and is often used in drain
openers. In order for commercial lye to work properly in soap, it must be pure sodium hydroxide, which is becoming difficult to find in grocery stores. If you are not sure if your lye is pure, you can look at the crystals. If you see flecks of gray or black, you should avoid it when making soap. You can also ask the manufacturer. Sodium Hydroxide is more powerful that Potassium Hydroxide so its SAP (saponification) value is different. The weight of Sodium is 40/56.1 as opposed to Potassium. |
|
| Useful Resources! |
| Soap Making Safety Procedures |
| Lye Calculator™ |
| Make Lye in a plastic bucket |
| Your 'Basic' Lye Machine | |
| This illistration is from the Jan/Feb 1972 issue of MotherEarth News. |
 |
| Lye in a pair of Plastic
buckets. |
 |
| Rough Wood bin Lye maker. The 'V' is made from Shiplap boards. The two ends are solid with on side haveing a small hole drilled thru to catch the liquid lye. |
 |
|
In crystal form, lye is easier to store and it also works well for many soap recipe measurements. Drying the lye can be accomplished by placing the lye water in the sun and allowing the water content to evaporate. As with the lye water, use caution with the lye crystals. Remember, do not use a metal
container. |
|
| Two Sample Soaps | ||
| Lard (Lye) Soap | Castille Soap | |
| 16 oz Lard 2.06 oz lye 4 – 6 oz water |
|
|
|
WARNING: Always add your solid form lye, sodium hydroxide or potassium hydroxide, to the liquid. If the liquid were added to the solid form lye a violent reaction could result.
This means you could have a "volcano" erupt out of your container.
Believe me you DO NOT want this to happen! |
||
| Basic Soapmaking Instructions | ||
|
||
 |
||
| Saponification Table | |||||||
| You take the weight of your Fat x the SAP factor to give you the weight of Lye for a batch of soap. | |||||||
| Base fat | SAP (Potassium) | SAP (Sodium) | Hard/Soft | cleansing | fluffy lather | stable lather | skin care |
| lard | 0.1945 | 0.1387 | hard | good | no | yes | fair |
| tallow | 0.1971 | 0.1405 | hard | good | no | yes | fair |
| soybean oil | 0.1906 | 0.1359 | soft | good | no | yes | fair |
| olive oil | 0.1898 | 0.1353 | soft | good | no | no | great |
| castor oil | 0.1804 | 0.1286 | soft | fair | yes | yes | great |
| coconut oil | 0.2680 | 0.1911 | hard | great | yes | no | fair |
| palm oil | 0.1992 | 0.142 | hard | great | no | yes | fair |
| peanut oil | 0.1921 | 0.137 | soft | fair | no | yes | great |
| jojoba oil | 0.0975 | 0.0695 | soft | fair | no | yes | great |
